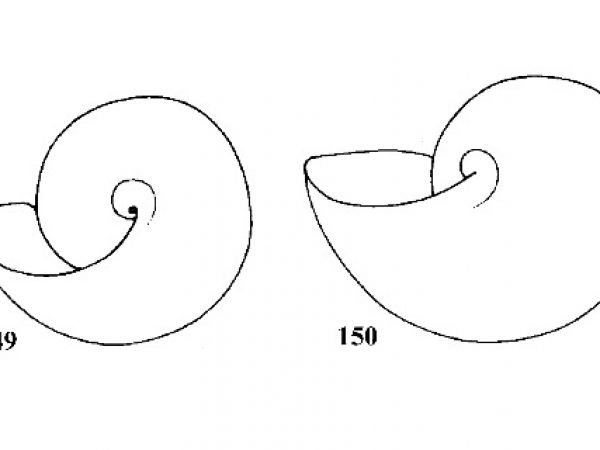
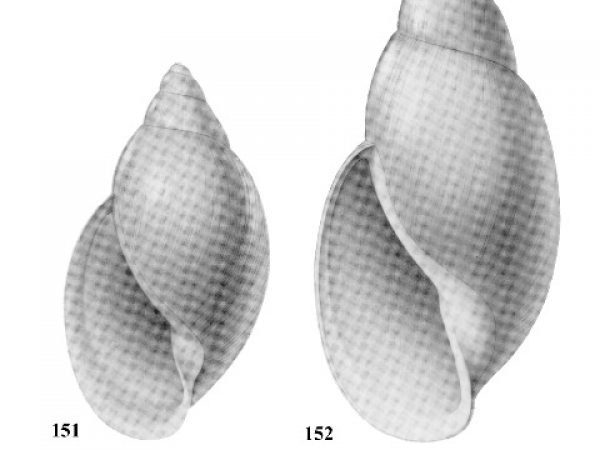
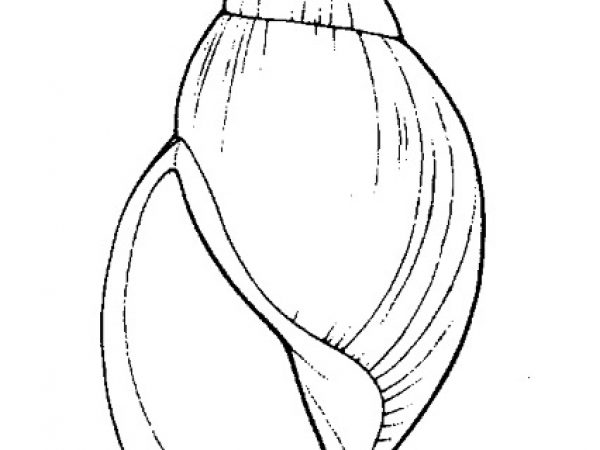
How to Use this Key:
- Start with the first question. Decide whether the statement in the first box (1a) or the second box (1b) best describes the characteristics of the snail you are trying to identify.
- Click on the link in the row that best matches your snail. Your choice will lead you to the proper identification of your snail or the next appropriate pair of questions.
- Work through the questions, each time choosing the characteristic that best matches your snail from the two choices. This will lead you to the final choice which identifies the snail.
- Click on images to enlarge them. You can also navigate through the images by clicking on the arrows near the left and right edges of the enlarged view.
| 1(a) | Aperture of shell closed by an operculum (snail must be collected live because the operculum is lost soon after death). Mantle cavity with gill on dorsal surface. | Question 2 |
| 1(b) | Aperture non-operculate; mantle cavity modified into a lung. | Question 59 |
| 2(a) |
Operculum calcareous with two pegs on inner surface (Figs. 1, 2). Aperture relatively large with fine, uneven, serrate denticles along parietal wall (Fig. 3). |
ID: NERITIDAE |
| 2(b) | Operculum corneas, without pegs on inner or outer surface. Aperture without serrate denticles on parietal wall. | Question 3 |
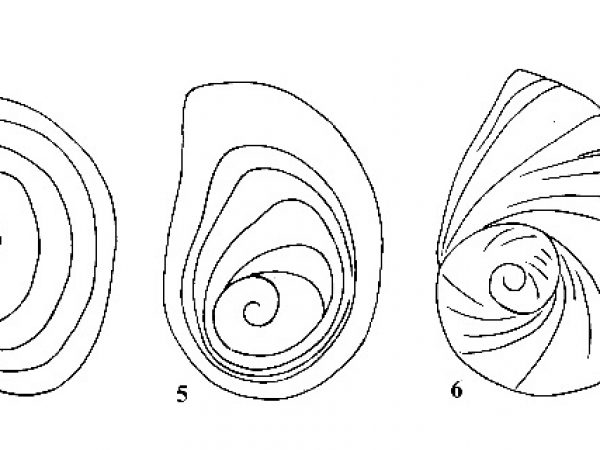
| 3(a) |
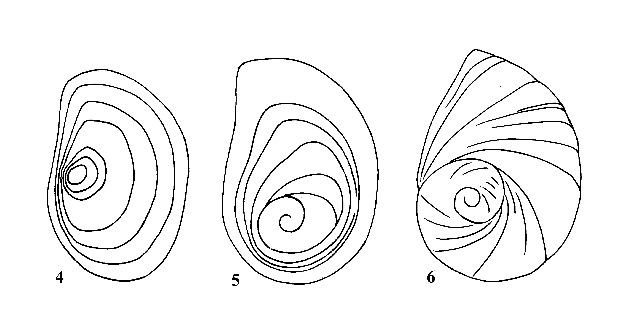 Operculum with concentric growth rings around nucleus (Figs. 4, 5). Shell medium to large (12-75 mm). |
Question 4 |
| 3(b) |
 Operculum spiral, consisting of two or more rapidly increasing whorls (Fig. 6). Shell usually small to medium (2-25 mm) |
Question 14 |
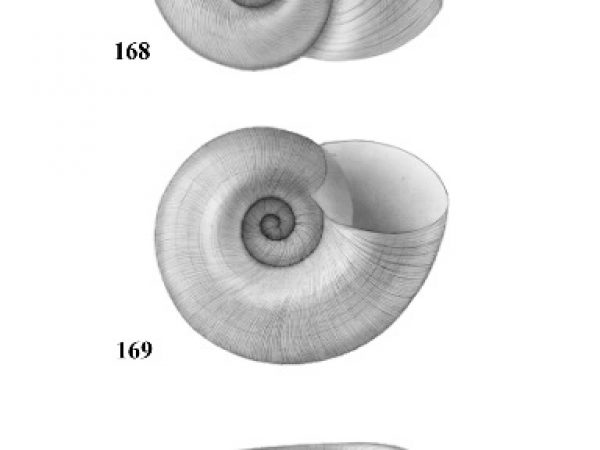
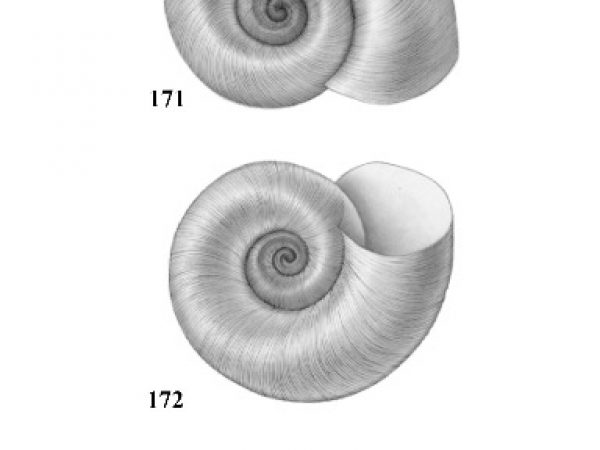
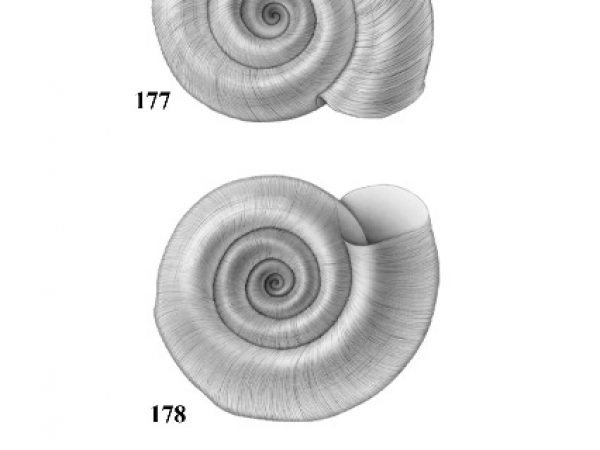
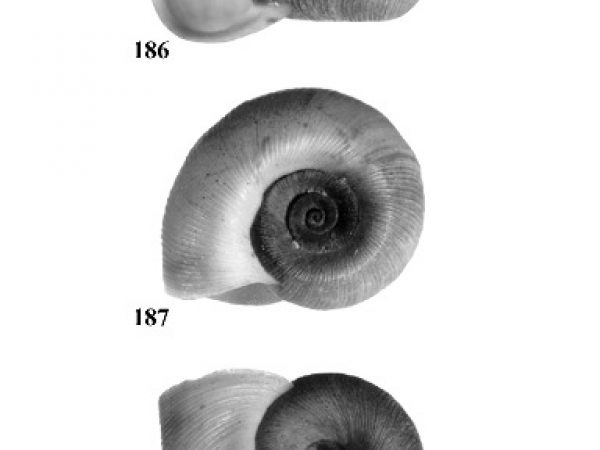
| 4(a) |
Umbilicus of shell perforated or broadly open. Spire depressed, much less than height of aperture, occasionally planular (Figs. 7-9). Aperture relatively ample (Figs. 10-12). In some genera, eggs are deposited above the water in dry clusters on stems, trees, walls, etc. In others they are deposited in the water in gelatinous clusters. Males with penis at anterior right corner of mantle cavity. |
Question 5 |
| 4(b) |
Umbilicus of shell closed. Spire usually about equal to or greater than height of aperture (Figs. 17-29). Females with live embryos in brood pouch in mantle; males with right eye stalk modified as a penis. |
Question 7 |
Family AMPULLARIIDAE
Medium- to large-sized tropical freshwater snails. One species occurs naturally in Florida, and three others have been introduced. The family contains many genera and numerous species in South America, Central America, Mexico, Africa, and Southeast Asia. Frequently the shells of ampulariids and viviparids are very similar. The shell characters given above for separating the families apply only to Florida species. The primary differences between the two families are based on soft anatomy. Ampullariids have been referred to as PILIDAE in earlier editions of this manual. The International Commision on Zoological Nomenclature has ruled that AMPULLARIIDAE is the valid name for this family of snails.
| 5(a) |
 Shell planispiral, adults large, 35-50 mm (Figs. 7-9). |
ID: Goldenhorn Marisa |
| 5(b) | Shell oval in shape. | Question 6 |
| 6(a) |
Whorls of spire pointed and scalariform (steplike with nearly flat shoulders and deep sutures). Apical whorls raised to form a point on top of spire. Adult shells about 40-60 mm high (Fig. 10). |
ID: Spiketopped Applesnail |
| 6(b) |
Apical whorls pointed and raised, but not scalariform. Shell large, 80-100 mm wide. Suture deeply impressed, forming a channel. Umbilicus wide (Fig. 11). |
ID: Channeled Applesnail |
| 6(c) |
Whorls of spire strongly arched or rounded. Apical whorls bluntly rounded and not conspicuously elevated. Adult shells about 40 – 70 mm high (Fig. 12). |
ID: Florida Applesnail |
Family VIVIPARIDAE

Medium to large freshwater snails found on all continents except South America. The family attains its greatest diversity in Southeast Asia. Two subfamilies occur in North America. The LIOPLACINAE is endemic and include Campeloma, Lioplax, and Tulotoma. The VIVIPARINAE is widely distributed throughout Europe, Asia and eastern North America. It is represented in North America by Viviparus. Various species of Cipangopaludina also have been introduced from the Orient into North America. One species, C. chinensis malleatus (Reeve, 1863) (Fig. 14), was introduced into a fishpond in St. Petersburg about 1921 and into lakes in Orlando about 1940. The current status of these introductions is not known.
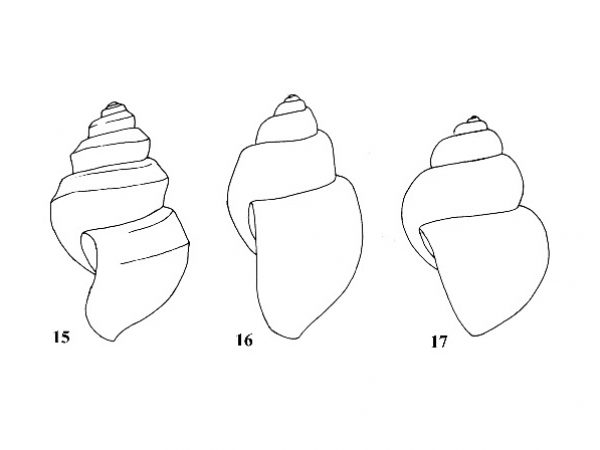
| 7(a) |
Nucleus of operculum spiral and then surrounded by concentric growth rings (Fig. 5). Outer lip of aperture strongly sinuous in lateral profile (Fig. 15). |
Question 8 |
| 7(b) |
Operculum constructed entirely of concentric rings (Fig. 4). Outer lip of aperture nearly straight in lateral profile (Figs. 16, 17). |
Question 9 |
Genus Lioplax — Troschel, 1856
A single species consisting of two highly variable subspecies occurs in Florida. Three other species occur farther north.
| 9(a) |
Shell thick and solid; whorls weakly convex or flattened (Figs. 16, 22-28). Shell unicolor, never banded. |
Question 10 |
| 9(b) |
Shell generally thin but strong; whorls of spire strongly convex with deep suture (Figs. 17, 29-32), shell usually banded in Florida forms. |
Question 13 |
Genus Campeloma — Rafinesque, 1819
Moderately large, thick-shelled operculate snails. Some species are dioecious with a normal representation of males and females. In others, some or all populations may be parthenogenetic, consisting only of females, or they may have a disproportionately small number of males. In parthenogenetic organisms each population is inbred in the strictest genetic sense, and frequently a population will have minor characteristics that distinguish it from others. There is no consensus in systematic biology about how to treat these forms. The criterion of inbreeding for defining species cannot be applied, and other objective criteria are not yet been established.
In Florida four types of Campeloma have been recognized. Campeloma geniculum (Conrad, 1834) is readily recognized by its obese, solid shell; it usually has a normal proportion of males in its populations, although some populations are apomictic parthenogens. The other three forms, C. limum (Anthony, 1860), C. floridense Call,1866 and C. parthenum Vail, 1979, are exclusively apomictic parthenogens and have been separated on the basis of aperture coloration, embryonic shell coloration, and contour of the outer lip (Vail 1979a). The coloration of the aperture and the embryonic shell is not consistent within single population samples, and the contour of the outer lip is highly variable within single drainage systems and with the age of the specimens examined. In view of the inconsistency of these shell characters, these three forms may represent only a single species, Campeloma limum, which is widely distributed from the Escambia River system of Florida and Alabama east and north into North Carolina and Virginia. More data based on many population samples from throughout this range will be necessary before the validity of the three forms can be determined. For the purpose of completeness, the three parthenogenetic forms and C. geniculum are included in the key.
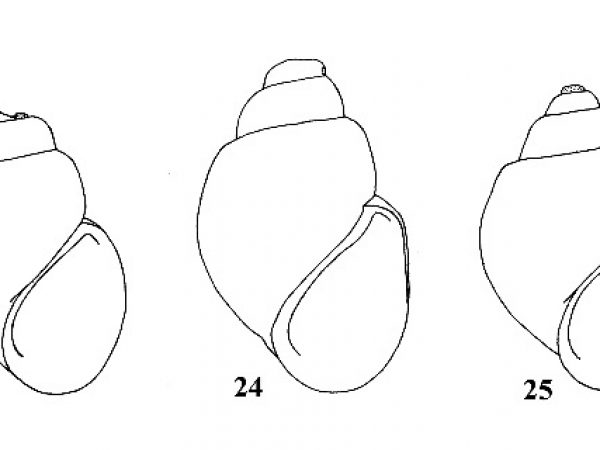
| 10(a) |
 Shell obese and ponderous. Spire short and compact, about 0.5-0.7 times height of aperture in mature specimens, proportionally longer in juveniles (Fig. 22). |
ID: Ovate Campeloma |
| 10(b) | Shell more slender and attenuate; spire more elongate in adults, about 0.7- 1.0 times height of aperture. | Question 11 |
| 11(a) |
Interior of adult aperture with brownish tinge. Newborn shells brown. Outer lip continuously rounded (Figs. 23, 26). |
ID: Purple-throated Campeloma |
| 11(b) | Interior of aperture livid white. Newborn shells white. Outer lip partially flattened in adults. | Question 12 |
| 12(a) |
Outer lip nearly straight and parallel to axis of shell in adults (Fig. 24, 27). |
ID: Maiden Campeloma |
| 12(b) |
Outer lip straightened just below shoulder; straightened portion lying at an angle to axis of shell (Figs. 16, 25, 28). |
ID: File Campeloma |
| 13(a) |
Shell ovate in shape, about 1.2-1.5 times as high as wide. Shell relatively thick (Figs. 16, 29). |
ID: Banded Mysterysnail |
| 13(b) |
Shell globose with a relatively depressed spire; about 1.0- 1.2 times as high as wide. Suture of whorls more deeply impressed than in previous species. Shell relatively thin. Color uniform olive-green, never banded (Fig. 32). |
ID: Rotund Mysterysnail |
| 14(a) |
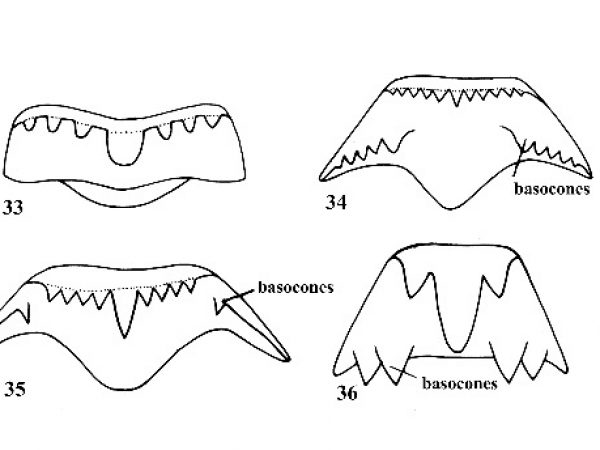
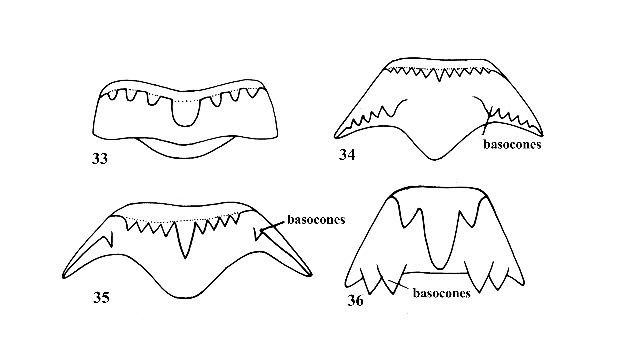 Shell elongate-conical; 10-40 mm long in adult specimens; apex of spire usually eroded; shell with 8-12 whorls although eroded specimens may have fewer; shell usually strongly sculptured with spiral and/or vertical ribs and threads (except in Elimia dickinsoni); central tooth of radula without basal or lateral cusps (Fig. 33); males without copulatory structures. |
Question 15 |
| 14(b) |
Shell variable in shape, elongate to globose; usually not more than 5 mm in length; apex of spire seldom eroded (except in Somatogyrus); 4-6 whorls; shell smooth (except in Pyrgophorus and Tryonia); central tooth of radula with basal cusps (Figs. 34, 35) (Pomatiopsis, Fig. 36); males with a copulatory appendage, a penis (Fig. 37), which is important for generic and specific diagnosis. (The penis is normally recurved into the mantle cavity, except during mating. |
Question 14 |
| 15(a) |
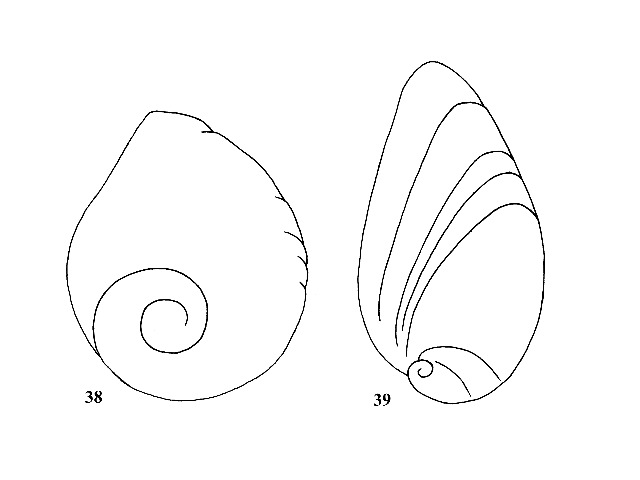 Operculum paleomelanian, with a large sub-centrally located nucleus (Fig. 38). (This character separates only the genera under consideration; genera from other areas of the world do not conform to this distinction.) Females with an egg-laying groove or sinus on right side of foot; eggs deposited on substrate. |
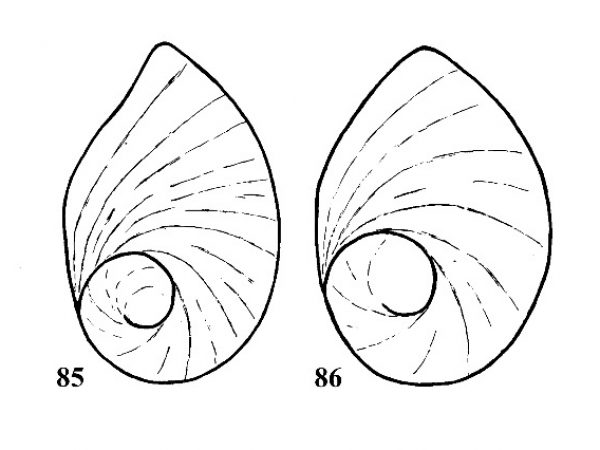
Question 18 |
| 15(b) |
 Operculum neomelanian, with a small, rapidly expanding nucleus located near the basal margin (Fig. 39). Primarily parthenogenetic; females viviparous with young snails in a brood pouch in nape. |
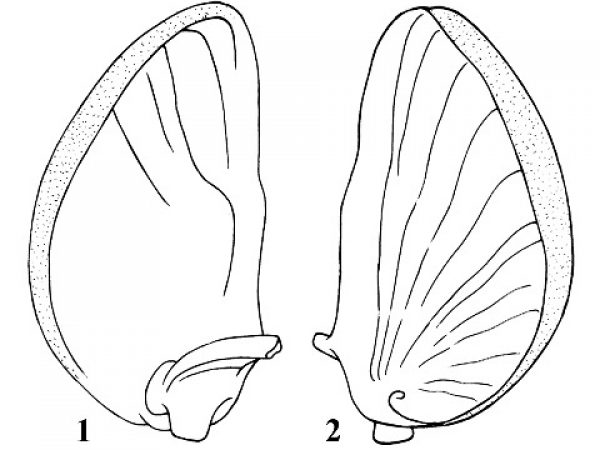
Question 16 |
Family THIARIDAE
Thiarids are found in tropical and subtropical regions of the world. Most inhabit fresh water, but some also occur in brackish water. The greatest diversity of genera and species is in the Indo-Australian region. Endemic New World genera occur in Cuba, Jamaica, and northern South America. Three species in Florida were introduced from Southeast Asia. Two, Melanoides tuberculata and Tarebia granifera, are medically important because they can serve as first intermediate host for the human lung fluke, Paragonimus westermani. Thiarids are ecologically significant because they tend to replace native snails where they are introduced and because of their abundance. In 1976 in the St. Johns River 1 found Melanoides tuberculatus with population densities of 10,000/m2.
| 16(a) |
Shell sculptured with prominent nodes overlapping suture and forming crenulations. Base of last whorl with prominent spiral ridges. Shell unicolor brown, never mottled with reddish spots. Shell about 0.38-0.41 times as wide as high and about 20-27 mm long. Sides of spire concave in outline (Fig. 40). |
ID: Quilted Melania |
| 16(b) | Shell sculptured with fine spiral striations, and in some cases curved axial ribs. Suture simple, not crenulated. Shell usually marked with red or rust-colored spots, flames, or bands (most apparent on juvenile specimens). Shell more slender, about 0.30-0.35 times as wide as high. Sides of spire straight-sided in outline. | Question 17 |
| 17(a) |
Shell sculptured with vertical, weakly curved ribs and much finer spiral striations. Prominent vertical ribs on middle and upper whorls. Base of shell with dark red spiral band. Shell light brown and frequently mottled with rust-colored spots that may form a spiral row below the suture. Attains a length of about 30-36 mm (Figs. 41-43). |
ID: Red-rimmed Melania |
| 17(b) |
Shell sculptured with fine spiral threads. Fine vertical ribs present on uppermost whorls. Base of shell usually without spiral band. Shell olive-green with vertical reddish flames and spots. Attains a length of 30-40 mm (Fig. 44). |
ID: Fawn Melania |
Family PLEUROCERIDAE
This family contains twelve genera in North America. Additional genera occur in the Orient, Southeast Asia, Africa, and South America. Only Elimia is found in Florida. Over 500 species of Elimia have been described, most on the basis of shell characters. Many have been synonymized, some undeservingly so. Biochemical studies show that in Elimia shell characters are conservative indicators of genetic divergence (Mihalcik & Thompson, 2002). Overshadowing this genetic divergence are frequent examples of convergent evolution of similar adult shell characters among distantly related species. This causes problems in species identification and yield an undermeasure of the actual number of species that exist in nature. Fortunately in Florida, the species are relatively easy to distinguish.
| 18(a) | Last whorl of adult shell smooth or with growth striations, but not with ribs and spiral chords. | Question 19 |
| 18(b) | Shell with prominent ribs and spiral chords on all whorls. | Question 20 |
| 19(a) |
Earlier whorls of adult shell with vertical ribs and spiral chords. Sides of spire straight in lateral profile. Last few whorls in large adults scalariform. Embryonic shell with fine vertical ribs and a heavy spiral chord on periphery and two basal spiral chords (Fig. 45). Adult large, about 23-27 mm long (Figs. 49, 50). |
ID: Stately Elimia |
| 19(b) |
Earlier whorls of adult shell with heavy vertical ribs crossed by knobby folds or a strong spiral chord; sides of spire straight or weakly convex in lateral profile; embryonic shell with a single spiral chord on periphery and heavy vertical ribs, but without basal spiral chord (Fig. 46). |
Question 20 |
| 20(a) |
Spire of adults with heavy vertical ribs; unicolor dark brown; usually less than 18 mm long (Figs. 51, 52). |
ID: Graphite Elimia |
| 20(b) | Spire of adults without distinct vertical ribs. | Question 21 |
| 21(a) |
Shell brightly banded and with large, wide and wavey vertical folds; robust, usually 21-25 mm long (Fig. 53). |
ID: Choctawhatchee Elimia |
| 21(b) |
Shell with or without bright bands; with low wavy growth wrinkles; large but not robust, 23-28 mm long. (Fig. 54). |
ID: Escambia Elimia |
| 21(c) |
Shell with or without bands; slender; Suture deeply impressed; upper whorls with a strongly carinate periphery (Fig. 55). |
ID: Dented Elimia |
| 22(a) | Shell with spiral chords or spirally arranged series of nodes. | Question 21 |
| 22(b) |
Shell with strong vertical ribs on spire; spiral chords vestigial, confined to spiral series of knobs on top of vertical ribs; adults small, about 13-16 mm long (Fig. 56). |
ID: Goblin Elimia) Elimia vanhyningiana (Goodrich, 1921). |
| 23(a) |
Shell unicolor, with distinct vertical sculpture in addition to strong spiral sculpture. Embryonic shell with a strong peripheral spiral chord that continues onto following whorls (Fig. 47). |
Question 24 |
| 23(b) |
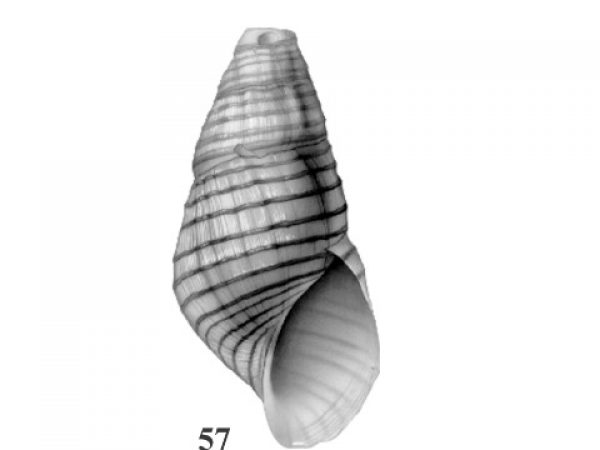
Shell with darker colored spiral chords that may be smooth or wavy. Vertical sculpture reduced to irregularly spaced and uneven growth striations or low undulating ribs. Embryonic shell smooth, without spiral chords (Fig. 48). Rather stocky, adult about 15-20 mm long (Fig. 57). |
ID: Black-crested Elimia) Elimia albanyensis (Lea, 1864). |
| 24(a) |
Sculpture above periphery of whorls consisting of nodes arranged in vertical and spiral rows. Shell short and stocky. Adult size small, about 12-16 mm long (Fig. 58). |
ID: Knobby Elimia |
| 24(b) | Sculpture above periphery of whorls consisting of sharp spiral chords and distinct vertical ribs; shell attenuate. | Question 25 |
| 25(a) |
Whorls straight-sided, not scalariform; suture hardly distinct; aperture terminating at periphery of last whorl; vertical ribs weaker and closely spaced; adult size about 25-30 mm long (Fig. 59). |
ID: Slackwater Elimia |
| 25(b) | Whorls of spire weakly scalariform, causing the suture to be deeply incised. Aperture terminating below periphery of body whorl. Vertical ribs relatively strong and more widely spaced. | Question 26 |
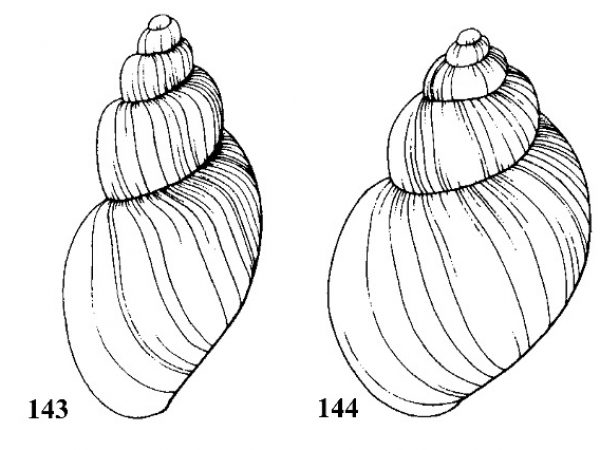
| 26(a) |
Vertical ribs smooth along the periphery, strongly developed (Fig. 60) |
ID: Waccasassa Elimia |
| 26(b) |
Vertical ribs smooth along the periphery, strongly developed (Fig. 60). |
Question 27 |
| 27(a) |
Vertical ribs strongly developed, crossed by nearly equal sized spiral threads that form low spines where they cross the ribs, adults about 20-25 mm long (Fig. 61). |
ID: Rasp Elimia |
| 27(b) |
Vertical ribs reduced in size, but with bold spines at the periphery; spiral threads above periphery relatively weak; adults up to 35 mm long (Fig. 62). |
ID: Spring Elimia |
| 28(a) |
Central tooth of radula with long basocones along base (Fig. 36). Amphibious; moves by step-like mode of progression (Fig. 63). |
ID: POMATIOPSIDAE |
| 28(b) |
 Central tooth of radula with basocones located on fore or lateral wing of tooth (Figs. 34, 35). Aquatic; moves by gliding motion. Shell of various shapes and sculpture. |
Question 29 |
Family HYDROBIIDAE
Hydrobiids are small- to medium-sized operculate snails that live primarily in brackish and fresh water. The HYDROBIIDAE include over 200 genera and approximately 1000 species. Most have monotonously simple, conical or depressed-conical shells that are nearly devoid of taxonomically useful characteristics. Essentially identical shells occur repeatedly among unrelated genera and subfamilies. Evolution has proceeded primarily through reproductive and trophic specializations. Important diagnostic characteristics for subfamilies, genera, and species are found in the female reproductive system, the male reproductive organ (the penis), and modifications of the radular teeth. The shell are secondarily useful for identification, but only when supplemented with anatomical information. The number of subfanilies recognized in the Hydrobiida is unsettled. Recent studies treat amnicolids as a separate families.
The author has attempted to simplify the key to include the minimum anatomical data needed for accurate identification. In the event that only shell specimens are available, picture-matching may be necessary to eliminate some choices in the couplets. Identification should conform with all data in the key and with geographic distributions. Only genera that enter fresh water are treated. Brackish-water genera, Onobops and Heleobops, that are common in Florida are omitted. Critical Florida references are Thompson 1968, 1969, 2000; Thompson and Hershler, 1991, and Hershler and Thompson, 1992.
Most freshwater hydrobiids in the eastern United States are annual species. They complete their life cycles in one year. Some species that are endemic to Florida springs live in environments with nearly constant temperatures and apparently reproduce continuously throughout the year. Other species deposit eggs during the period of March through May, at which time the adults die and immature forms dominate the population through August or September. Many samplings of hydrobiids collected during the period of May through September are not identifiable because only immature forms are present, and important diagnostic anatomical characteristics have not yet developed.
| 29(a) | Shell globose with a short depressed spire; body whorl ample; umbilicus closed. | Question 30 |
| 29(b) | Shell conical or cylindrical-conical in shape; spire elevated; body whorl not disproportionately voluminous; umbilicus closed or open. | Question 32 |
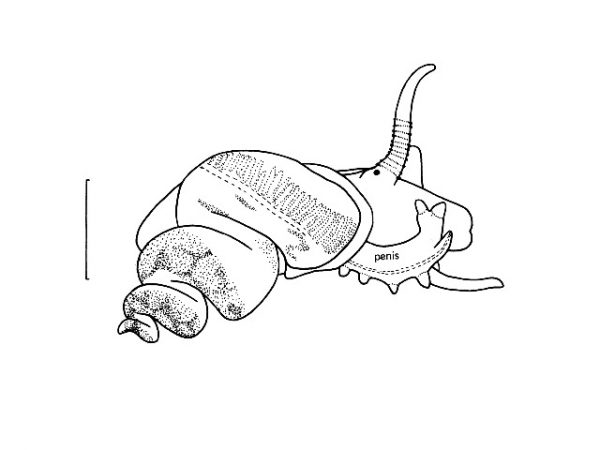
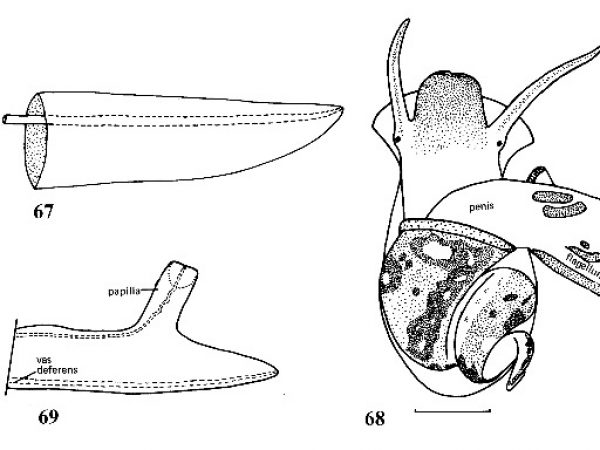
| 30(a) |
Columellar margin of aperture wide, flat-faced; apex of spire usually erroded; apical whorls, when present, with minute spiral striations; central tooth of radula with basocones located on ridged surface of tooth (Fig. 34); penis frlattened, blade-like (Fig. 67). |
Question 31 |
| 30(b) |
Columellart margin of aperture not flat-faced; apex of shell usually entire; penis with large terminal lobe on left side and small appendage-like flagellum on right margin (Fig. 68); lobe and flagellum with various patterns of dermal glands; central tooth of radula with basocones located on reflected margin of tooth (Fig. 35). |
Question 48 |
Genus Somatogyrus — Gill, 1863
Somatogyrus is distributed from the Mississippi River drainage system east to Atlantic coastal streams, and south to the Gulf coastal drainage systems. Approximately 35 species have been described. Three species are known from rivers entering northwest Florida from Georgia and Alabama.
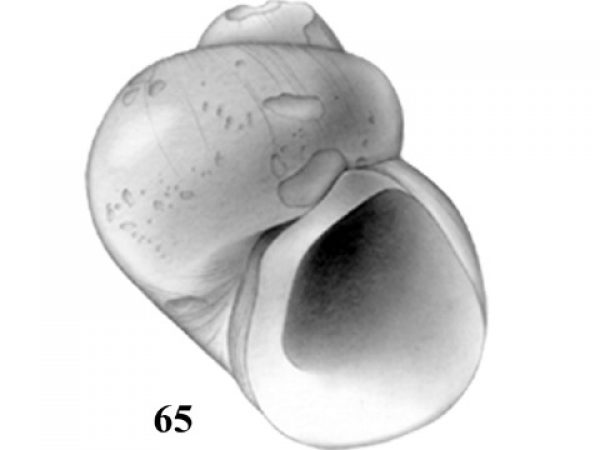
| 31(a) |
Adult shell about 4-5 mm high; umbilicus wide; columellar margin of the aperture concave in outline (Fig. 64). |
ID: Gulf Coast Pebblesnail) Somatogyrus walkerianus (Aldrich, 1905). |
| 31(b) |
Adult shell small, less than 3 mm high; umbilicus narrow but not occluded by the columellar lip; columellar margin of the aperture weakly concave (Fig. 65). |
ID: Choctawhatchee Pebblesnail) Somatogyrus sp. |
| 31(c) |
Adult shells small, less than 3 mm high; umbilicus closed by the reflected columellar lip; columellar margin of the aperture nearly straight, barely convex (Fig. 66). |
ID: Apalachi Pebblesnail |
| 32(a) |
Penis of males with papillae along sides (Figs. 69, 70). |
Question 33 |
| 32(b) |
 Penis with large a terminal lobe on the left side and a smaller appendix-like flagellum on the right side. Lobe and flagellum with various patterns of dermal glands (Fig. 68). |
Question 48 |
| 33(a) |
 Penis with a single papilla along left margin (Fig. 69). Papilla with a duct that leads to a blind caecum in nape of snail. Vas deferens independent of blind caecum and its duct. |
Question 64 |
| 33(b) |
Penis with papillae along both margins, or right margin only (papillae absent in some Aphaostracon). Vas deferens the only duct present in penis (Fig. 70). |
Question 34 |
| 34(a) |
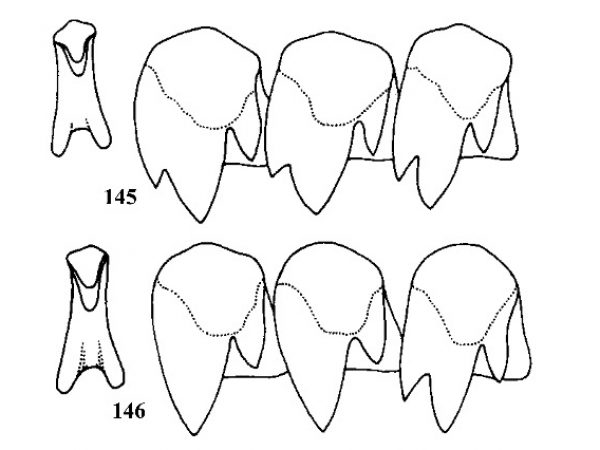
Shell usually with raised spiral threads around periphery, frequently the uppermost thread has conical or triangular spines (Figs. 72-74). Female ovoviviparous with about 50 embryos in uterus; embryos clearly evident through clean shells. Penis with 3-7 papillae along right margin and a projection with 1-4 papillae near end on left side (Figs. 70, 71). |
ID: Serrated Crownsnail |
| 34(b) | Shell without conical spines, although spiral threads may be present. Penis with different arrangement of papillae than above. Females ovoviviparous or oviparous. | Question 35 |
| 35(a) |
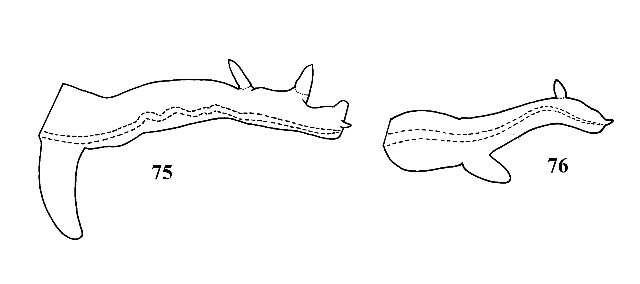 Shell elongate, thin, transparent, grayish. Suture deeply impressed. Female ovoviviparous with about 15 large embryos in uterus. Penis with large papilla on right margin near base and one or two papillae on left margin near distal end (Figs. 75, 76). Shell sexually dimorphic in size, males about one-third as long as females. |
Question 36 |
| 35(b) | Shell variable in shape. Female oviparous, never with embryos in uterus. Penis without a conspicuously enlarged papilla near right base, although other papillae usually are present. | Question 37 |
| 36(a) |
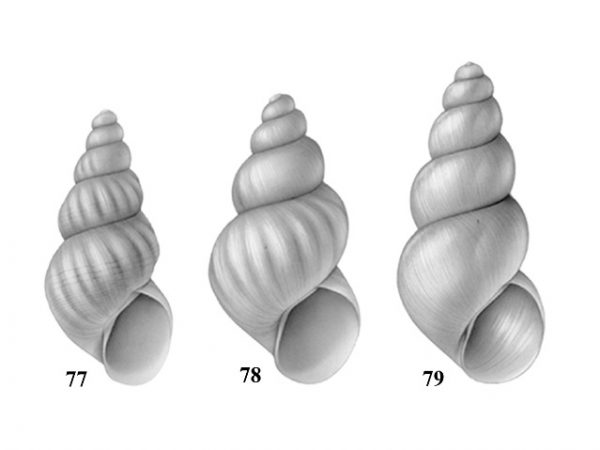
Shell elongate-conical, with about 5-7 whorls. Aperture broadly in contact with preceding whorl. Peristome incomplete around aperture. Umbilicus closed or narrowly perforate. Spiral or costate sculpture usually present. Female shell about 4-6 mm long (Figs. 77-79). Penis with 2 or 3 papillae on left margin (Fig. 75). |
ID: Smooth-ribbed Hydrobe |
| 36(b) |
Shell cylindric-conical with 4.5-5.0 whorls. Aperture free from, or only in slight contact with, preceding whorl. Peristome complete around aperture. Umbilicus open. Shell sculpture with incremental striations only. Female shell about 3.5-4.0 mm long (Fig. 80). Penis with one papilla on left margin (Fig. 76). |
ID: Regal Hydrobe |
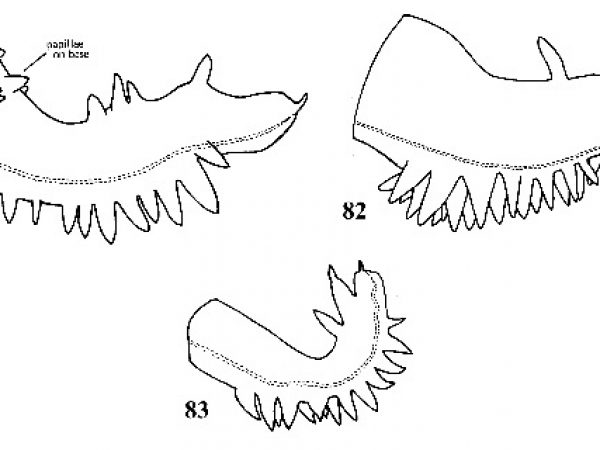
| 37(a) |
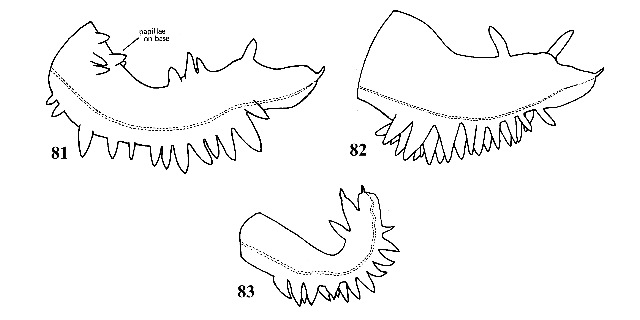 Shell conical, olivaceous in color. Peristome incomplete around aperture. Whorls flat-sided with suture weakly impressed. Sculptured with fine incremental striations and a few fine spiral striations (difficult to distinguish except with transmitted light). Sexes not conspicuously dimorphic in size. Penis with 7-50 papillae along right margin and 1-4 papillae along distal third of left margin (Figs. 81-83). |
Question 38 |
| 37(b) |
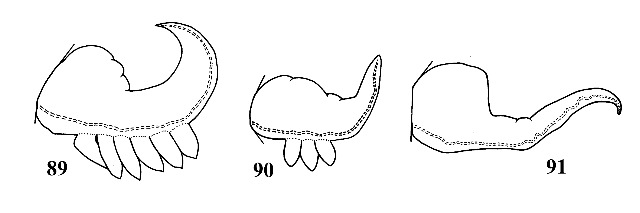 Shell conical or cylindric-conical; light to dark brown. Peristome complete around aperture. Whorls generally arched. Spiral sculpture absent. Sexes strongly dimorphic in size, males about half as long as females. Penis with 0-6 papillae confined to right margin (Figs. 89-91). |
Question 40 |
Genus Littoridinops — Pilsbry, 1952
Littoridinops is found most frequently in brackish water, although the three species found in Florida also occur in frehswater. The genus is found along the east coast of North America from Maryland south to Tabasco, Mexico.
| 38(a) |
Penis with 3 – I 0 small papillae around base and 7-15 papillae along right margin in a single row (Fig. 81). Sides of spire slightly convex. Parietal margin of operculum concave (Fig. 85). Shell with a brownish hue. Shell with 5.0-6.5 whorls; about 3-5 mm long (Fig. 84). |
ID: Henscomb Hydrobe |
| 38(b) | Penis without papillae around base. | Question 39 |
| 39(a) |
Penis with 17-50 papillae along right margin arranged in 3-5 rows (Fig. 82). Parietal margin of operculum slightly convex in outline (Fig. 86). Shell highly variable in shape; freshwater forms olivaceous or brown; adults with about 4.5-6.0 whorls; about 3.5-4.5 mm long (Fig. 87). |
ID: Cockscomb Hydrobe |
| 39(b) |
Penis with 5-15 papillae along right margin arranged in 1-2 rows (Fig. 83). Parietal margin of operculum convex. Shellolive-colored; spire straight-sided. Shell with 5.6-6.8 whorls; about 3.2-4.5 mm long (Fig. 88). |
ID: Bantam Hydrobe |
Genus Aphaostracon — Thompson, 1968
This genus of minute freshwater snails is endemic to the Florida peninsula.
| 40(a) |
 Penis with papillae along right margin (Figs. 89, 90). |
Question 41 |
| 40(b) |
 Penis simple, elongate-conical, without papillae (Fig. 91). |
Question 42 |
| 41(a) | Penis with 1-2 papillae. | Question 44 |
| 41(b) | Penis with 3-5 papillae. | Question 46 |
| 42(a) |
Shell squat, compact, cylindric-conical in shape, thick and opaque. About 4.2-4.6 weakly arched whorls with a weakly impressed suture. Aperture comma-shaped, tightly appressed against preceding whorl. Length of shell about 2.2-2.8 mm long (Fig. 92). |
ID: Dense Hydrobe |
| 42(b) | Shell conical with strongly arched whorls and a deeply impressed suture. | Question 43 |
| 43(a) |
Shell translucent. Body whorl inflated. Aperture elliptical and loosely attached to or slightly separated from preceding whorl; 4.5-4.9 whorls. Length of shell 2.6-3.0 mm (Fig. 93). |
ID: Clifton Spring Hydrobe |
| 43(b) |
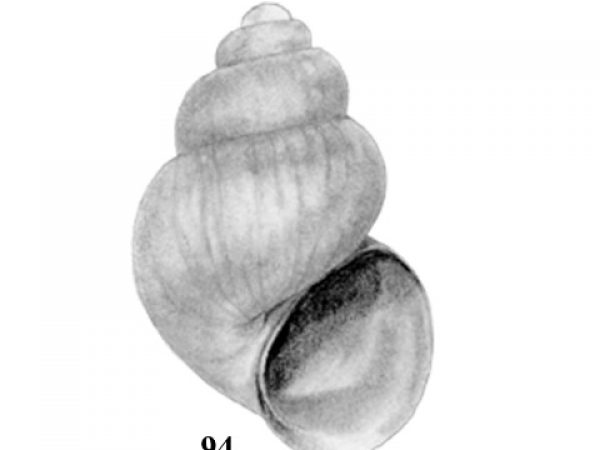
Shell very thin, fragile, transparent. Aperture elliptical and usually attached to preceding whorl; 4.1-4.5 whorls present. Length of shell 2.0-2.4 mm (Fig. 94). |
ID: Blue Spring Hydrobe |
| 44(a) |
Shell narrow, terete or cylindric-conical with a moderately impressed suture. Aperture broadly attached to preceding whorl across parietal wall. Whorls 4.6-5.3. Length of shell 2.4-3.4 mm (Fig. 95). |
ID: Slough Hydrobe |
| 44(b) | Shell conical to elongate-conical; whorls 4.0-4.5 with a deeply impressed suture. Aperture narrowly in contact or free from preceding whorl. | Question 45 |
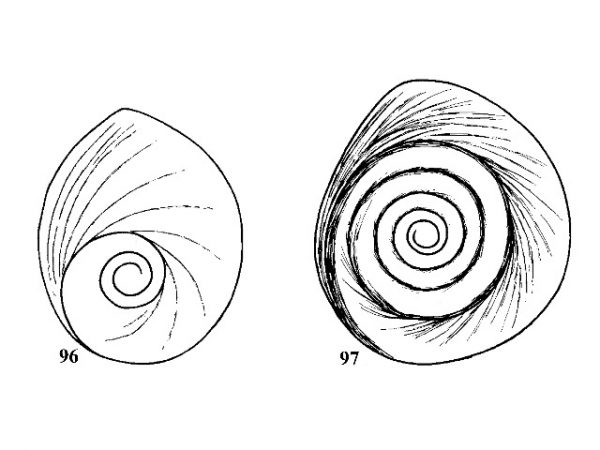
| 45(a) |
Shell conical to ovate-conical. Aperture broadly elliptical. Operculum with about 3 whorls (Fig. 96). Length of shell 2.0-2.4 mm (Fig. 98). |
ID: Suwannee Hydrobe |
| 45(b) |
Shell elongate-conical. Aperture broadly ovate. Operculum with about 6 slowly expanding whorls (Fig. 97). Length of shell 2.2-2.6 mm (Fig. 99). |
ID: Wekiwa Hydrobe |
| 46(a) |
Shell cylindric-conical, solid opaque; 4.5-4.9 moderately rounded whorls. Aperture ovate; broadly attached to preceding whorl. Length of shell 2.1-2.8 mm (Fig. 100). |
ID: Thick-shelled Hydrobe |
| 46(b) | Shell conical; thin and transparent; 4.0-4.5 strongly arched whorls. Aperture loosely attached to or widely separated from preceding whorl. | Question 42 |
| 47(a) |
Aperture loosely attached to or slightly free from preceding whorl. Length of shell 2.0-2.5 mm (Fig. 101). Penis with 3 papillae along right margin (Fig. 90). |
ID: Fenney Spring Hydrobe |
| 47(b) |
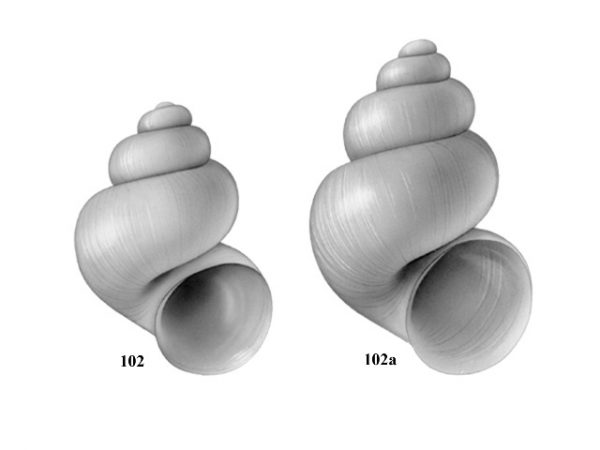
Aperture widely separated from preceding whorl. Length of shell 3.0-4.0 mm (Fig. 102a, 102b). Penis with 4-6 papillae along right margin (Fig. 89). |
ID: Freemouth Hydrobe |
Subfamily NYMPHOPHILINAE
This subfamily includes about fifteen North American and European genera (Thompson 1979). It is characterized by the lobed shape and glandular patterns of the penis. Evolution has occurred through reproductive specialization, with each genus and most species having distinctive patterns of dermal glands. Identifications are difficult in many instances without properly preserved specimens.
| 48(a) | Shell globose or tear-shaped. Umbilicus closed. | Question 49 |
| 48(b) | Shell conical or cylindric-conical. Umbilicus open, although very narrow in some species; occasionally closed. | Question 50 |
| 49(a) |
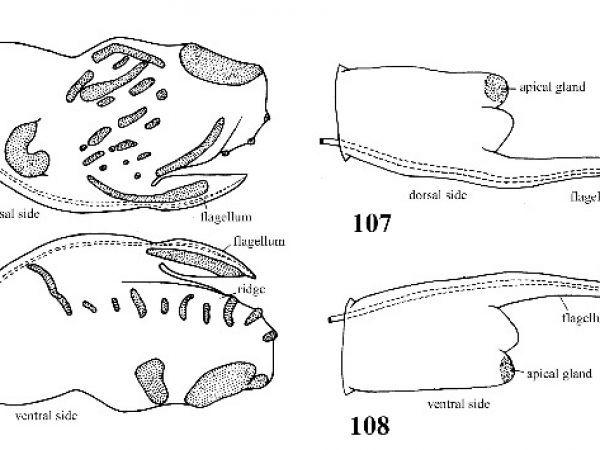
Shell medium-sized or large; 5.4-7.5 mm long; thick and opaque; ovate or globose; apical whorls depressed; sides of spire rounded; body whorl conspicuously enlarged, ample, rapidly descending to aperture along last half whorl. Columellar margin of aperture very thick but not reflected over umbilical area (Fig.103). Outer lip straight in lateral profiles. Penis with a small blade-like flagellum along right margin and a heavy mid-ventral ridge that bears 8-11 narrow transverse dermal glands; other glands present on terminal lobe and flagellum (Figs. 105, 106). |
ID: Alligator Siltsnail |
| 49(b) |
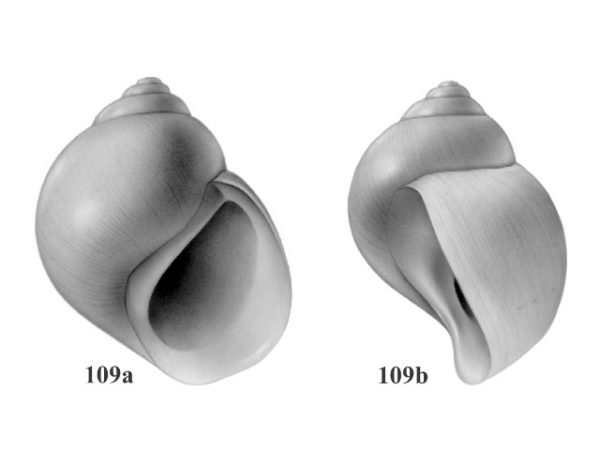
Shell medium-sized, 3.0-3.8 mm long; thinner, translucent or transparent in life; tear-shaped; spire raised and nearly straight-sided, pointed; body whorl less conspicuously enlarged, not descending to aperture along last half whorl. Columellar margin of aperture wide, rounded in front like a spindle. Outer lip of aperture arched forward (Figs. 109a, 109b). Penis with a very long slender flagellum that extends beyond terminal lobe. A single glandular crest present on apex of terminal lobe (Figs. 107, 108). |
ID: Teardrop Snail |
| 50(a) |
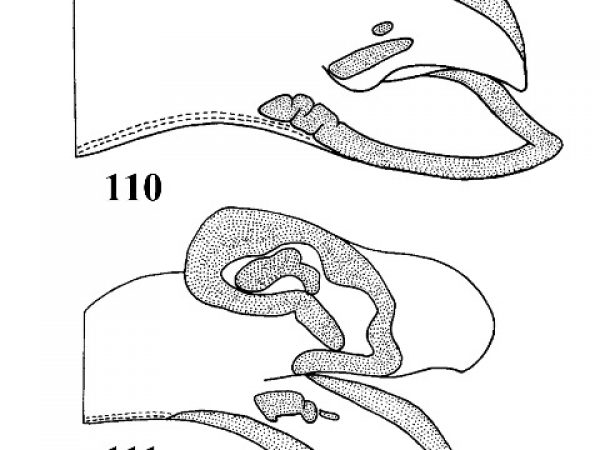
Mantle richly marked with reddish-brown blotches and spots. Terminal lobe of penis slender. Penis with a large, blade-like flagellum with continuous heavy dermal glands along each side (Figs. 110, 111, 68). |
Question 51 |
| 50(b) |
Mantle spotted with black, shaded or unmarked; terminal lobe of penis with complex crests that cause it to look like an animal’s head; flagellum slender, with scattered and discontinuous glands along edge (Figs. 115, 116). |
Question 52 |
Genus Spilochlamys — Thompson

This genus contains three species. Two occur in Florida. One species, Spilochlamys turgida (Thompson, 1969), the Pumpkin Siltsnail (Fig. 112), occurs in the Ocmulgee River system in Georgia. The genus is difficult to diagnose by shell characters because the three species are very dissimilar. Spilochlamys is most similar to the next genus, Cincinnatia, from which its shell usually differs by having a more deeply impressed suture, more strongly shouldered whorls, and a wider umbilical perforation.
| 51(a) |
Umbilicus narrow, without a strong circum-umbilical ridge, outer lip without a callus on inner surface. Penis as illustrated (Fig. 110). Shell with about 4.5-5.0 whorls; about 3.0-4.8 mm long (Fig. 113). |
ID: Conical Siltsnail |
| 51(b) |
Umbilicus widely perforate, accentuated by a strong circum-umbilical keel. Outer lip of aperture with a thick callus on inner surface. Penis as illustrated (Fig. 111). Shell with 4.4-5.0 whorls; about 3.7-5.2 mm long. (Fig.114). |
ID: Armored Siltsnail |
Genus Floridobia — Thompson & Hershler, 2002
In previous editions of the manual species of Floridobia were placed in the genus Cincinnatia. The latter genus includes a single species, Cincinnatia integra (Say, 1817). Floridobia is known from peninsular Florida north along the Atlantic seaboard to Maine.
| 52(a) |
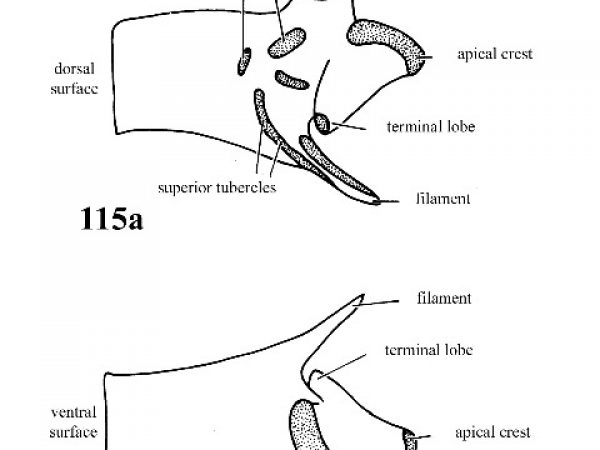
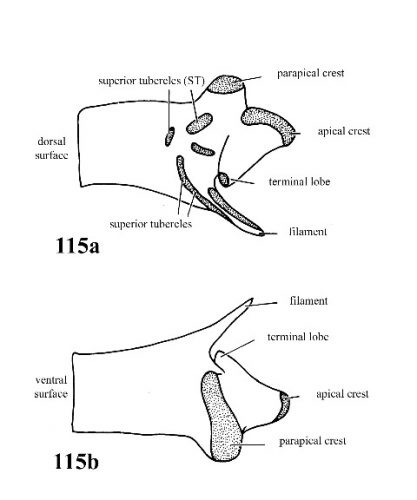 Penis with a few simple scattered superior tubercles on outer surface; parapical crest on a low ridge; accessory crest and inferior crest absent (Fig. 115a, 115b). |
Question 53 |
| 52(b) |
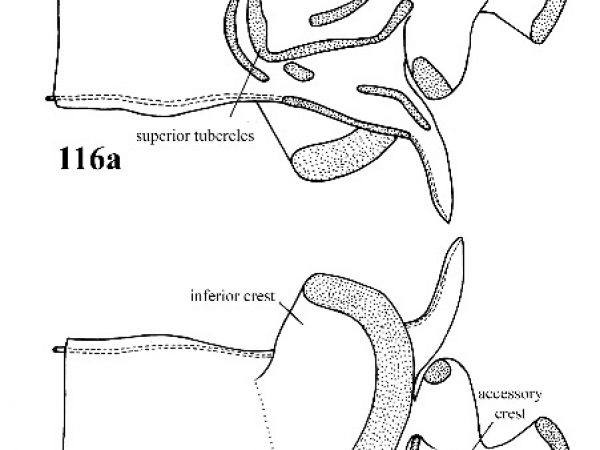
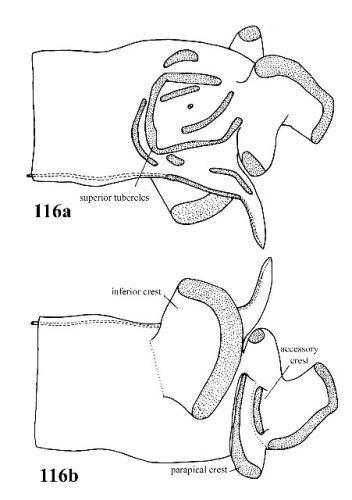 Penis with some superior tubercles fused into a U-shaped crest; parapical crest raised on a fleshy pedicel; accessory crest and inferior crest usually present (Fig. 116a, 116b). |
Question 58 |
| 53(a) |
Spire long, 1.3-1.7 times length of aperture, convex in outline; shell relatively large and nearly cylindrical; 4.0-4.9 mm long; whorls 5.0-5.7, strongly rounded with a deeply impressed suture (Fig. 117). |
ID: Crystal Siltsnail |
| 53(b) | Spire shorter, conical, 0.7-1.3 times length of aperture, nearly straight-sided in outline, suture of whorls weakly or moderately impressed. | Question 54 |
| 54(a) |
Shell minute, 2.0-2.3 mm long; thin and transparent; spire 0.7-1.1 times length of aperture; flagellum lacking glandular crests. shell with 3.9-4.2 whorls (Fig. 118). |
ID: Ichetucknee Siltsnail |
| 54(b) | Shell larger, thicker, and opaque; spire 0.9-1.3 times length of aperture; with more than 4.2 whorls; penis flagellum with glandular crests. | Question 55 |
| 55(a) |
Shell medium-sized, 2.8 – 3.5 mm long; spire 0.9 – 1.1 times length of aperture; shell with 4.2-4.7 whorls (Fig. 119). |
ID: Hyacinth Siltsnail) Floridobia floridana (Frauenfeld, 1863). |
| 55(b) | Shell moderately large, 3.7-4.5 mm long; spire 1.00-1.35 times length of aperture; shell with 4.8-5.4 whorls. | Question 56 |
| 56(a) |
Aperture broadly ovate in shape; parietal wall weakly in contact or solute from body whorl; whorls more prominently arched and with a deeper impressed suture. (Fig. 120). |
ID: Enterprise Siltsnail) Floridobia monroensis (Dall, 1885). |
| 56(b) | Aperture elliptical or trapezoidal in shape. | Question 57 |
| 57(a) |
Aperture elliptical in shape; spire slenderer and slightly convex in outline; suture moderately impressed suture; parapical crest reduced in size. (Fig. 121). |
ID: Flatwood siltsnail |
| 57(b) |
Aperture trapezoidal in shape; spire more robust, straight sided; with a shallower suture; parapical crest large (Fig. 122) |
ID: Alexander Siltsnail |
| 58(a) |
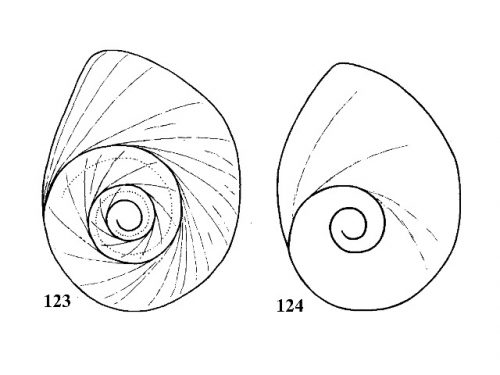 Operculum nearly multispiral with four large, slowly expanding whorls (Fig. 123). |
Question 59 |
| 58(b) |
 Operculum paucispiral, with about three rapidly expanding whorls (Fig. 124). |
Question 60 |
| 59(a) |
Shell small, about 3.0-3.5 mm long; adult with 4.4-4.8 whorl; lower corner of aperture tending to be angulate with fluted channel (Fig. 125); accessory crest absent on penis. |
ID: Seminole Siltsnail |
| 59(b) |
Shell large, 3.7-4.5 mm long; adults with 4.6-5.3 whorls; lower corner of aperture slightly extended forward as a weak, tongue-like projection (Fig. 126); accessory crest present on penis. |
ID: Rock Springs Siltsnail |
| 60(a) |
Shell minute, 2.4-2.7 mm long; adults with 3.9-4.2 whorls; spire short, 0.8- 1.1 times height of aperture (Fig. 127); superior tubercles on penis arranged in oblique longitudinal series. |
ID: Pygmy Siltsnail) Floridobia parva (Thompson, 1968). |
| 60(b) | Shell larger, 2.8-4.6 mm long; adults with 4.0-4.9 whorls; spire longer, 0.9-1.3 times height of aperture; superior tubercles not arranged in oblique longitudinal series. | Question 61 |
| 61(a) |
Shell large, 4.0-4.6 mm long; thick and opaque. Whorls globose, with a deeply impressed suture. Penis with two or more longitudinal crests within U-shaped superior tubercle (Fig. 128). |
ID: Ponderous Siltsnail |
| 61(b) | Shell 2.8-3.7 mm long. Thin and translucent or transparent. U-shaped superior crest not enclosing longitudinal crests. | Question 62 |
| 62(a) | Shell conical, spire moderatly high. Suture relatively deep, Whorls of spire rounded. Penis with invaginated cave-like pit within U-shaped superior tubercles. | Question 63 |
| 62(b) |
Shell elongate conical, spire high. Suture relatively shallow. Whorls of spire less rounded. Penis with superior tubercles forming a vague broken loop; invaginated cave-like pit absent. Parapical crest of verge greatly enlarged. Penis filament white. (Fig. 129). |
ID: Creek Siltsnail |
| 63(a) |
Penis with a dense pattern of superior tubercles. Inferior crest absent. Accessory crest present. Penis filament white. (Fig. 130). |
ID: Wekiwa Siltsnail |
| 63(b) |
Dorsl surface of penis with a few elongate superior tubercles that form a weakly defined U-shaped pattern. Accessory crest absent. Inferior crest usually present. Penis filament black. (Fig. 131). |
ID: Green Cove Springsnail |
Family AMNICOLIDAE
This family includes four North American genera, Amnicola, Dasyscia, Colligyrus and Lyogyrus, and the European genus Marstoniopsis and some Asian genera.
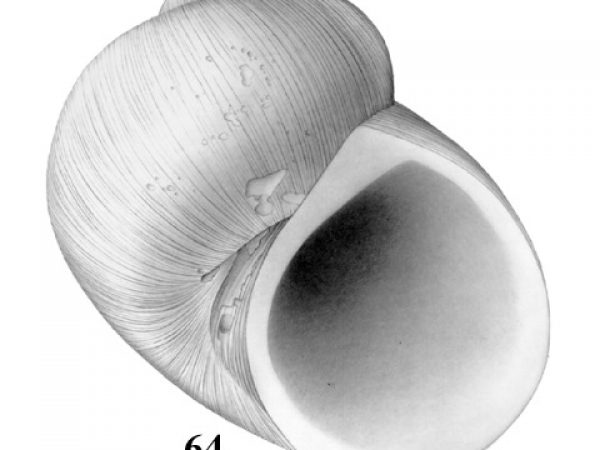
| 64(a) | Shell small, 2.0-2.8 mm long. Nuclear whorl slightly protruding, 0.29-0.41 mm in diameter transverse to initial suture. Mantle diffusely pigmented or unpigmented. | Question 65 |
| 64(b) | Shell larger, 2.2-4.8 mm long. Nuclear whorl flattened, 0.41-0.48 mm in diameter transverse to initial suture. Mantle mottled with black spots and blotches. | Question 66 |
Genus Amnicola — Gould, 1841
Amnicola is a widespread genus in temperate North America. Four species currently are recognized, but the genus has received very little study over most of its range. The specie prefer quite clear water, of lakes and backwaters of streams andsprings.
| 66(a) |
Shell conical, thick, opaque. Umbilical perforation wide, 1/6 to 1/8 diameter of shell. Whorls 3.8-4.3. Incremental striations intensified near nucleus and umbilicus. Aperture rhomboid; baso-columellar angle extended as slight tonguelike projection; basal lip broadly but shallowly indented (Fig. 135). |
ID: Squaremouth Amnicola |
| 66(b) | Shell conical or globose-conical; thin, translucent, occasionally opaque. Umbilical perforation narrow, 1/8 – 1/10 diameter of shell. Whorls 3.0-4.0. Incremental striations uniformly weak. Aperture ovate; baso-columellar angle not extended; basal lip not indented. | Question 67 |
| 67(a) |
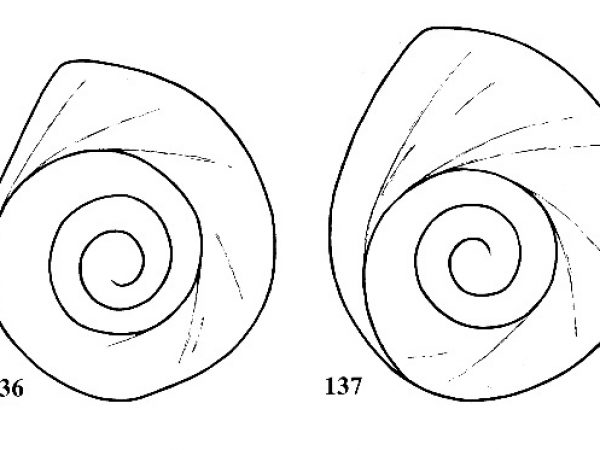
Operculum tightly coiled with four slowly expanding whorls (Figs. 136, 138). |
ID: Amnicola dalli. dalli |
| 67(b) |
Operculum loosely coiled, with 3.75 large whorls, the outer one expanding more rapidly than the others (Figs. 137, 139). |
Amnicola dalli johnsoni (Pilsbry, 1899). |
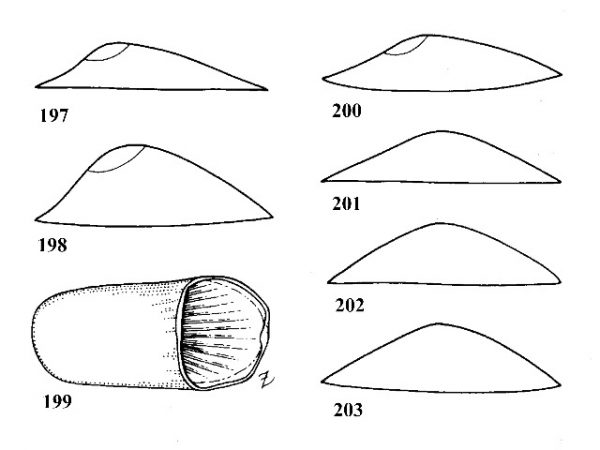
| 68(a) |
Shell cap-shaped or limpet-like with a wide, open aperture forming base of shell (Figs. 197-209). |
Question 88 |
| 68(b) | Shell coiled. | Question 69 |
| 69(a) | Shell planular or disc-shaped; spire flat when raised above succeeding whorls (Figs. 159-196). |
Question 78 |
| 69(b) | Shell elongate with pointed conical spire. | Question 70 |
| 70(a) |
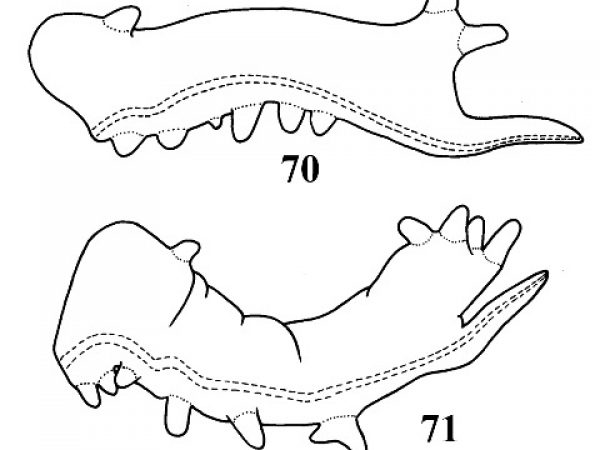
Shell coiled to the right, with the aperture on the right side (Figs. 140-146). |
Question 71 |
| 70(b) |
Shell coiled to the left, with the aperture on the left side (Figs.147-158). |
Question 73 |
Family LYMNAEIDAE
Lymnaeids are a nearly cosmopolitan family of freshwater snails. They achieve their greatest diversity in genera and species in temperate North America. The lymnaeid fauna of the southeast is particularly depauperate compared to more northern and western regions of the continent. Only three widely distributed species occur in Florida.
| 71(a) |
Aperture large, oval, much more than half the length of shell. Sculpture consisting of distinct spiral striations and threads superimposed on growth striations (Fig. 140). |
ID: Mimic Pondsnail |
| 71(b) | Aperture relatively shorter, about half or less the length of the shell. Sculpture consisting of incremental striations only. | Question 72 |
Genus Fossaria — Westerlund, 1883
Fossaria is found throughout North America, and in northern Europe. It contains about a dozen species in North America. The species are highly variable, and there is no consensus regarding the number of valid species.
| 72(a) |
Aperture narrow, about half as wide as high. Whorls shouldered above near suture (Fig. 143). Body whorl compressed (Fig. 141). Radula with tricuspid lateral teeth (Fig. 145). |
ID: Rock Fossaria |
| 72(b) |
Aperture oval, about 0.75 times as wide as high. Whorls uniformly rounded, not flattened above (Fig.144). Body whorl rounded (Fig. 142). Radula with bicuspid lateral teeth (Fig. 146). |
ID: Carib Fossaria |
Family PHYSIDAE
Taylor (2003) monographed the family Physidae. This monumental work changed all previous concepts concerning subfamilies, genera and species in the family. Taylor recognizes twenty-three genera based on the soft anatomy. In previous editions of the manual the Florida species were placed in the genus Physella. This revision follows the classification proposed by Taylor. The reader may be troubled by the imprecise shell characteristics that are used in the key. Physid species have similar and superficially featureless, variable shells, and the shell is imprecise for identifying most genera and many species. Definitive identifications may require anatomical dissections and reference to other literature.
| 73(a) |
Apex very short, only slightly raised above body whorl. Size small I I to medium, 8-12 mm in length. Sculpture consisting of axial striations only (Fig.147). |
ID: Low-dome Physa |
| 73(b) | Apex proportionally longer, about 0.3-0.5 times length of shell. | Question 74 |
| 74(a) |
Shell elliptical-ovate in shape. Apex distinctly convex in outline. Shell usually opaque in adults, with a dark red callus inside the lip (Fig. 148). |
ID: Tadpole Physa |
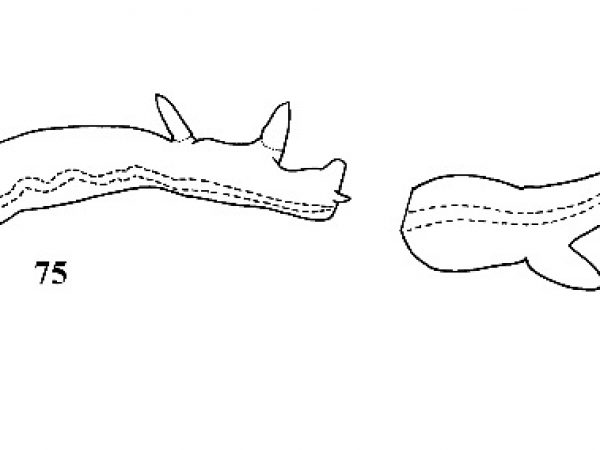
| 74(b) | Shell generally elliptical in outline. Apex nearly straight-sided or concave in outline. Shell transparent or opaque. Inside of lip with or without a reddish callus but callus never dark red in color. | Question 75 |
| 75(a) |
Shell slender, attenuate. Apex about 0.4-0.5 times length of shell. Suture weakly impressed. Base of shell open when viewed from below, showing most of preceding whorl due to curved basal lip (Fig. 149). Surface smooth, glossy, striations when present very weak. Amber to milky white in color (Figs. 151, 152). |
ID: Bayou Physa |
| 75(b) |
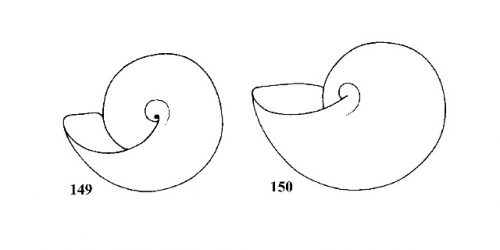 Apex less than 0.4 times length of shell. Suture more deeply impressed. Base of shell when viewed from below showing about half of preceding whorl due to straight basal lip (Fig. 149). Sculpture variable. Color of fresh shell never milky white. |
Question 76 |
| 76(a) |
Shell usually large, about 12-16 mm long. Generally with distinct axial and spiral striations. Brownish-yellow in color (Fig. 153). |
ID: Bayou Physa |
| 76(b) | Shell smaller, generally less than 12 mm long. Spiral sculpture absent. Shell transparent or translucent. Gray to brownish-yellow in color. | Question 77 |
| 77(a) |
Shell elliptical in shape. Suture not as deeply impressed as in 77b. Last whorl not shouldered (Figs.155, 156). |
ID: Carib Physa |
| 77(b) |
Shell usually squarish or rectangular in shape. Suture more deeply impressed than in 77a. Last whorl distinctly shouldered. Texture dull. Axial striations distinct (Fig. 158). |
ID: Pewter Physa |
Family PLANORBIDAE
Planorbid snails are restricted to fresh water and occur worldwide, being found on all continental landmasses and many oceanic islands. Most species are disc-shaped or planular, as is implied by the name PLANORBIDAE. The basic shell morphology of the PLANORBIDAE is left-handed, or sinistral (FIGS. 180-193). Many species are secondarily modified to appear right-handed or pseudo-dextral (FIGS. 159-179). The family contains numerous genera and species, some of which are important medically as intermediate hosts for trematode parasites. Genera that serve as intermediate hosts for schistosomatid trematodes have been studied extensively. Most other genera have received relatively minor attention, and their systematics are in flux. Few of the North American species have been studied to the extent that their taxonomy, geographic distributions, life histories, and ecology are documented. The planorbid fauna of the southeastern states is particularly poorly known. The following key includes all of the species that occur in Florida and some that occur in Alabama, Georgia, and South Carolina.
| 78(a) | Size small, discoidal, adults seldom exceeding 4 mm in width. | Question 79 |
| 78(b) | Size larger, adults 7 mm or more in width. Shell usually planispiral, but two species may have a flat-topped, elevated spire. | Question 83 |
| 79(a) |
Shell keeled or strongly angular at the periphery (Figs. 161, 164, 167). Weak spiral striations present on top of growth striations (Figs. 159, 162, 165). Shell with three whorls. Umbilicus narrow (Figs. 160, 163, 166). |
Question 80 |
| 79(b) |
Shell rounded at the periphery (Figs. 170, 173). Spiral sculpture faint or absent (Figs. 169, 172). Shell with 3-4 whorls. Umbilicus variable. |
Question 82 |
| 80(a) |
Body whorl relatively narrow, not conspicuously enlarging near aperture (Fig. 159). Apex of shell slightly convex in outline. Body whorl strongly keeled as though pinched at the periphery (Figs. 159-161). |
ID: Marsh Sprite |
| 80(b) |
Body whorl relatively rapidly expanding in diameter (Fig. 162). Aperture enlarged (dilated). Apex of shell flat, though it may be raised above the periphery of the last whorl (Figs. 164, 167). Periphery variable. |
Question 81 |
| 81(a) |
Periphery of last whorl bluntly angular, lying below plane of apex (Figs. 162-164). |
ID: Bugle Sprite |
| 81(b) |
Body whorl angular. Last whorl flattened above. Apex suppressed to form a nearly flat plane with the peripheral angle (Figs.165-167). |
ID: Micromenetus dilatatus avus (Pilsbry, 1905). |
| 82(a) |
Shell dark brown. Umbilicus narrow, deep (Fig. 169). Body whorl uniformly rounded peripherally (Fig. 170). Aperture moderately oblique. Shell sculptured with very fine, uniformly spaced axial threads (Figs.168-170). Marginal teeth of radula spatulate and bearing 8-10 weakly differentiated cusps. |
ID: Penny Sprite) Micromenetus floridensis (Baker, 1945). |
| 82(b) |
Shell grayish-white. Umbilicus broad, shallow (Fig. 172). Periphery of body whorl below middle, side of whorl flattened and sloping toward apex (Fig. 173). Aperture strongly oblique. Shell nearly smooth, sculptured with irregularly spaced incremental striations (Figs. 171-173). Marginal teeth of radula trapezoidal with well-differentiated cusps. |
ID: Ash Gyro |
| 83(a) |
Adults with six teeth in aperture, two on the parietal wall and four on the palatal wall. Apical whorls depressed; body whorl obtusely angular above and sharply carinated around funnel-shaped umbilicus. Lip of adult shell usually with a thick crest externally and an internal callus. Adults about 6-8 mm wide (Figs. 174-176). |
ID: Thick-lipped Rams-horn |
| 83(b) | Adult shell without lamella on interior walls of aperture. | Question 84 |
| 84(a) |
Shell discoidal, grayish-white in color; transparent when fresh. Shell nearly smooth, sculptured with irregular growth striations. Adults with about 5 whorls, and 10-13 mm wide (Figs.177-179). |
ID: Ghost Rams-horn |
| 84(b) | Shell usually corpulent, brown or green, generally opaque, but occasionally translucent in juveniles. | Question 85 |
| 85(a) |
Shell distinctly carinated above and below; both apex and base funnel-shaped. Sculpture consisting of fine incremental threads, and spiral striations that may become obsolete at maturity. Adults 9-15 mm wide (Figs. 180-182). |
ID: Two-ridged Rams-horn |
| 85(b) | Whorls rounded, not carinate above; occasionally angular below. Apex of shell flat-topped, but sunken below periphery of last whorl. Base of shell regularly expanding, not funnel-shaped. | Question 86 |
| 86(a) |
Shell dull. Sculptured with regularly spaced, fine, incremental threads and spiral striations; spiral striations evident only on base of whorls in some specimens. Apex flat with narrow threadlike keel. One to several dark growth-rest varixes usually present on last whorl. Adults about 15-18 mm wide with about 5.5 whorls (Figs.183-185). |
ID: Marsh Rams-horn |
| 86(b) | Shell glossy. Surface smooth or dented with hammer marks. | Question 87 |
| 87(a) |
Shape highly variable, usually disc-shaped but some specimens with flat-topped raised spire. Lower margin of aperture advanced beyond upper margin so that plane of aperture slopes posteriorly when viewed from the side (Figs.189-193). |
ID: Seminole Rams-horn |
| 87(b) |
Spire raised and flat-topped. Lower margin of aperture not advanced beyond upper margin. Plane of aperture nearly vertical when viewed from the side. Size smaller than last species, seldom exceeding 10 mm in width (Figs.194-196). |
ID: Mesa Rams-horn |
Family ANCYLIDAE
Ancylid gastropods are small, fragile limpets found in most freshwater habitats. All freshwater limpets in the southeast belong to this family. Other families occur elsewhere in North America and in other continents. Little is known about these animals, and diverse opinions exist as to how many species are recognizable. Many North American snail species were described near the turn of the century by Walker (1918). Basch (1963) revised the classification and recognized only twelve species. However, this appears to be an over reduction, and several southern species were synonymized that appear worthy of recognition. Six species are known to occur in Florida. They are variable in their shell characteristics, and some samples may be difficult to identify. The following key is modified from Basch (1963). It is essential that clean specimens be examined under proper lighting to observe diagnostic sculpture characteristics.
| 88(a) |
Shell usually elevated, but variable. Apex with fine radial striations (Figs. 204-207), often eroded in older specimens. Peristome narrow to broadly ovate. Aperture usually open; occasionally with a horizontal, shelf-like septum closing posterior part (Fig. 199). |
Question 89 |
| 88(b) |
Shell depressed. Apex smooth, with no trace of radial striations (Fig. 208, 209), although radial striations may be present on other areas of shell. Peristome ovate to subcircular. Aperture never with a septum. |
Question 90 |
Genus Ferrissia — Walker, 1903
Ferrissia is widely distributed in the northern hemisphere. About fifteen species have been described from North America. Basch (1963) recognized five valid species. There is much disagreement as to how many species actually exist. Two species occur in Florida.
| 89(a) |
Shell thin, fragile, very much depressed, less than 0.25 times as high as long. Color often glossy reddish brown. Apex fairly prominent as a rounded knob in the right posterior quadrant (Figs. 197, 204). Length of shell up to 5 mm. |
ID: Hood Ancylid |
| 89(b) |
Shell moderately elevated, over 0.25 times as high as long. Apex in the posterior right third, domelike and evenly rounded. Adults about 2-3 mm long (Figs. 198, 205). |
ID: Blackwater Ancylid |
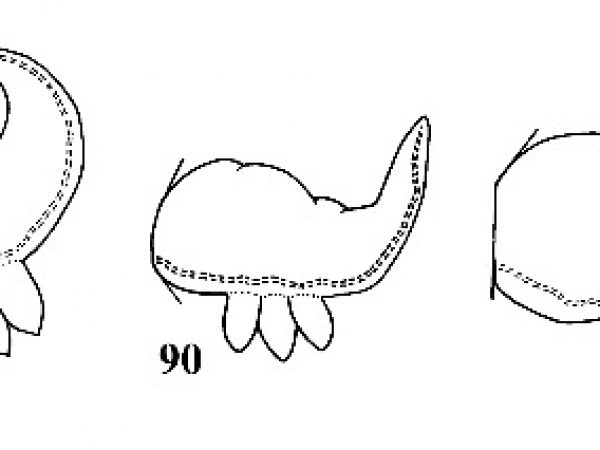
| 90(a) | Apex very obtuse, almost in the middle of the shell. Radial striations present or absent Laevapex Walker, 1903. | Question 91 |
| 90(b) |
Apex subacute, distinctly eccentric to the right of the midline. Radial striations present on shell but not on apex. Average length about 4 mm (Figs. 200, 206). |
ID: Excentric Ancylid |
Genus Laevapex — Walker, 1903
Laevapex is a North American genus. Eight species have been proposed. Three occur in Florida. They are commonly found on live aquatic plant stems, dead leaves and sticks, and on bottles and cans.
| 91(a) |
Shell elongate. Radial riblets on entire surface of shell except apex. Apex slightly behind and slightly to the right of midline. Adults about 5 mm long (Figs. 201, 207). |
ID: Peninsula Ancylid |
| 91(b) | Shell ovate or subcircular in shape, smooth or with radial sculpture localized anteriorly. | Question 92 |
| 92(a) |
Shell ovate, smooth or with fine raised riblets; riblets usually on the anterior slope when present. Apex behind center of shell. Average length about 5 mm (Figs. 202, 208). |
ID: Dusky Ancylid |
| 92(b) |
Shell subcircular, smooth, often encrusted with dark material. Apex in about middle of shell. Average length about 5.5 mm (Figs. 203, 209). |
ID: Cymbal Ancylid |